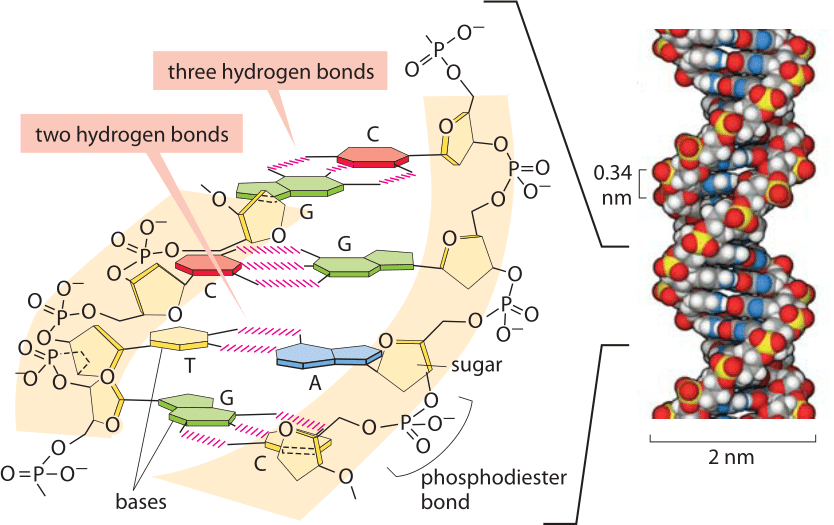
Dotted bonds are going back into the screen or paper away from you, and wedge-shaped ones are coming out towards you. Note: The solid line represents a bond in the plane of the screen or paper. The molecules which have this extra bonding are: These relatively powerful intermolecular forces are described as hydrogen bonds. In the cases of NH 3, H 2O and HF there must be some additional intermolecular forces of attraction, requiring significantly more heat energy to break. If you repeat this exercise with the compounds of the elements in Groups 5, 6 and 7 with hydrogen, something odd happens.Īlthough for the most part the trend is exactly the same as in group 4 (for exactly the same reasons), the boiling point of the compound of hydrogen with the first element in each group is abnormally high. Note: If you aren't sure about van der Waals dispersion forces, it would pay you to follow this link before you go on. The increase in boiling point happens because the molecules are getting larger with more electrons, and so van der Waals dispersion forces become greater. If you plot the boiling points of the compounds of the Group 4 elements with hydrogen, you find that the boiling points increase as you go down the group. Many elements form compounds with hydrogen. If you are also interested in the other intermolecular forces (van der Waals dispersion forces and dipole-dipole interactions), there is a link at the bottom of the page. It is suggested that the precise alignment of these hydrogen bonds contributes to stability of the double helix and ensures the proper alignment of the corresponding base pairs.This page explains the origin of hydrogen bonding - a relatively strong form of intermolecular attraction. The hydrogen bonds are formed between the oxygen atoms (red) and the adjacent N-H bonds, and between the central nitrogen (blue) and the adjacent N-H bond. According to the Watson-Crick model, the double helix of DNA is assembled and stabilized by hydrogen pairing between matching “bases”. Hydrogen bonding is also important is DNA. The extra energy that is required is necessary to break down the hydrogen bonding network. Although these two compounds have similar molar masses, a significant amount of energy must be put into the polar molecule, water, in order to move into the gas phase, relative to the non-polar methane. Water, with a molar mass of 18, has a boiling point of +100 ˚C. Methane, CH 4, has a molar mass of 16 and a boiling point of –164 ˚C. For many compounds which do not possess highly polarized bonds, boiling points parallel the molar mass of the compound. Hydrogen bonding is generally used to explain the high boiling point of water (100 ˚C). Even though hydrogen bonds are relatively weak, if you consider that every water molecule is participating in a least four hydrogen bonds, the total energy of hydrogen bonding interactions can rapidly become significant. O hydrogen bond is about 5 kcal/mole (21 kJ/mole), or less than 5% of the energy of a “real” covalent bond.The energy required to break the O-H covalent bond (the bond dissociation energy) is about 111 kcal/mole, or in more proper SI units, 464 kJ/mole. Hydrogen bonds are weak, relative to covalent bonds. Recall that oxygen has two unshared pairs in its valence shell, and the hydrogen-oxygen interaction in water is the classic example of a hydrogen bond. A bond that is formed from a hydrogen atom, which is part of a polar covalent bond (such as the O-H bond) to another, more electronegative atom (that has at least one unshared pair of electrons in its valence shell) is called a hydrogen bond. By this logic, it is not at all surprising that water can also react strongly with itself, and indeed water exists as a vast network of molecules aligned so that their positive and negative dipoles interact with each other. We attributed this to the ability of water molecules to align themselves so that the polarized hydrogen-oxygen bonds could stabilize cations, anions, and virtually any compound that also contained a significantly polarized covalent bond. In Chapter 7, we explored the unique properties of water that allow it to serve as a powerful solvent with the ability to dissolve both ionic compounds, as well as polar molecular compounds.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
